Category Archives: Academic Chemistry
Connecting the Characteristics of Life
Lab Safety… it’s no Accident
Today we’ll be going over some important information about your safety in a Lab setting. After review the Lab Safety rules (check the documents page for your specific class for a PDF version), take a look at the image below. See if you can spot some incidences of failing to follow lab procedures for safety.

Reminder there will be a “Common Sense” Lab Safety Quiz tomorrow and you must pass with 100% before being allowed to use the lab.
Also! You must have a signed Lab Safety Contract turned into Mr. Kubuske by Wednesday to be allowed to use the lab.
Solutions for Solutions
Haha, I get it. Seriously though, we’re going to take some baby steps through the world of solutions. As a refresher, Solutions are a homogenous mixture (meaning it is evenly dispersed) of Solute and Solvent. The solvent is always the substance of larger amount when the two are mixed…
For example, the Earth’s atmosphere is made up of several different types of gases. It is only about 20% oxygen but 76.5% Nitrogen! That being said, we would say that Nitrogen is the Solvent and all other gases are the solutes.
There are different types of mixtures as well.
- Collids – are also Homogeneous, but the molecules of a collide do not settle or separate (like Milk)
- Suspensions – are heterogeneous meaning the parts of the mixture are not evenly dispersed (think Italian Dressing)
When it comes to solutions though, its a complete and even mixture. But not everything mixes so well. In chemistry, we say that “Like dissolves Like“. What that means is that things that have a smiler molecular make-up can mix together, like salt and water. we call these substances “miscible” because they are each polar molecules so they can mix. But Oil, which is non-polar and there for not like water, does not mix with water. We call substances that do not mix “immiscible“. But fear not oil fans, oil can mix with other non-polar substances because they are alike!
So what does mix with water? Well good solutions of water include Electrolytes. Electrolytes are compounds that can be broken down, or separated into ions. Salt, NaCl, is an electrolyte because it is split into Na+ and Cl– when placed in water. Non-electrolytes can also mix with water, but they do not separate. So solid (s) sugar molecules, since it cannot be split into ions, will stay as one molecule but will change form to become aqueous (aq). Why is that important? Well we calculate the number of particles created when a molecule is placed in solution with the V’Hoff factor (i). For example, Sodium Hydroxide, which is an electrolyte, dissociates (that means molecule made with ionic bonds is broken into ions) into Na+ and OH–. The V’Hoff factor would be i=2 because it splits into one of each of the ions. If it was MgCl2 , then i=3 because there would be one Mg+2 and two Cl–. For all Non-electrolytes, i=1 because the molecule does not separate. Like when sugar is placed in water… the sugar does not break apart, it stays together as C6H12O6. If it’s a covalenetly bonded molecule splitting into ions, it is called ionization.
What if we wanted to make Kool-aid and want to know exactly how much to place in 100g of water before it starts to just settle at the bottom. Then we need a Solubility curve…  Each line represents a different compound. Where the line does shows saturation, or the maximum amount of solute that can be dissolved in 100g of water before it starts to settles at the bottom. For Example, at 50°C we can dissolve 30g of K2Cr2O7 in 100g of H20. But at 90°C we can dissolve 69g of K2Cr2O7. We can also use this graph to determine if a solution is ‘supersaturated‘ (more than saturated – above the line) or ‘unsaturated‘ (less than saturated – below the line).
Each line represents a different compound. Where the line does shows saturation, or the maximum amount of solute that can be dissolved in 100g of water before it starts to settles at the bottom. For Example, at 50°C we can dissolve 30g of K2Cr2O7 in 100g of H20. But at 90°C we can dissolve 69g of K2Cr2O7. We can also use this graph to determine if a solution is ‘supersaturated‘ (more than saturated – above the line) or ‘unsaturated‘ (less than saturated – below the line).
For every solution there are two mole measurements we can determine. These measurements can help us determine how much solute or solvent is in a solution. These measurements are:
- Molarity (M) = molessolute/Literssolution
- Molality (m) = molessolute/kgsolvent
For these calculations, you can convert between moles and grams by either multiplying by molar mass (mol x MM = g) or by dividing by molar mass (g/MM = mol). If you need, click this link for a molar mass calculator. Luck There is so much more to study with solutions(colligative properties, ppm, & % solution), this is just the tip of the iceberg.
Thursday 4/24

You’re reaction when you see Mr. Kubuske is not here today.
I’m really sorry for having to be out again. I had a feeling I might have to be, which is one of the reasons I pushed the Bio test back to Monday ad allowing retakes on Monday in Chem. You’ve got a fairly straight-forward day ahead of you however. Here’s what I need you to get accomplished today.
Biology
- Email or Turn in your Energy in Food lab.
- Do the “Energy Transfer” review worksheet you received on monday. You can get the PDF version from the documents page as well. Turn it in when complete.
- Use the PPT of the Succession Notes (The video I made would not load properly to YouTube) and make drawings for Primary Succession & Secondary Succession. Be sure to write down key terms and a brief description of Eutrophication.
- Do the questions attached to the notes.

Would this abandoned amusement park be considered Succession? Would it be primary or Secondary?
Chemistry
- If you are ready to take the test, ask the Sub for a copy of the test and a formula sheet. You may NOT use your iPad, even as a calculator. There are calculators at your table and in the back of the room.
- If you are not taking the test today, make sure you have completed all of the 10 steps by tonight because YOU WILL be taking the test tomorrow (you may retake it on Monday if needed).
Wednesday 4/16
Students, I hate to tell you bit I have to be out one more day to comfort my family and make my way back to Indiana.

In the mean time, I need you to take notes to answer these three questions for Biology:
- What impacts do human’s have on their environment?
- What causes ‘climate change’?
- What is an ‘invasive species’ and how is it harmful to an ecosystem?
Use the PPT provided, use your eBook, and/or watch the YouTube video on the topic to answer these questions. Tomorrow we’ll be playing a game related to the topic of Human Impacts on ecosystems and you’ll need these notes done to participate.
When you have the notes completed use the notes and pages 314-323 in your ebook to answer questions about 4 ‘case studies’ which can be used to show the principles of ecology. Your job will be to use the information on these pages to answer questions about each case study from the pdf below. The 4th case study, about invasive species, is found in the second pdf below.
- Case Studies Questions – Due Tomorrow
- Case Study #4 Invasive species
Please have these questions completed for class tomorrow.
Chemistry, please continue to work on Gas Law calculations and email me as you complete tasks.
Honoring a Hero

My Grandfather, Father, and Me (2012)
I’m sorry to say but I will not be with you in class on Monday or Tuesday. I’ll be in Ohio honoring my grandfather who passed away this weekend. He was a great man who defended his country, raised a family, and improved his community. He led a full happy life and he will be forever loved and missed.
Leroy E. Kubuske 1925-2014
So I’m going to leave some directions on what I need you to do for me while I’m gone so we can pick up right where we need to on Wednesday. Theres no handout because I did not have an opportunity to make any copies before i had to leave town. So if you want handouts they are available as pdf’s from the semester 2 documents page. Thanks for your help and understanding.
Biology
Over the next Two days please get the following items accomplished. Feel free to do them in any order you wish.
- Biome Books are Due Tuesday. Feel free to use Monday’s class to work on them.
- Cycles Notes. I need you to get information on three of the major biogeologic cycles (Carbon, Nitrogen, & Water). I have placed images for each of the cycles on the documents page as well as using the links below. Study the images and take notes on them as needed. When you are ready, answer the three questions below using information you discerned from the images.
- (Water) As I got a drink from the drinking fountain at school, my teacher told me I was drinking the same water a dinosaur did 65 million years ago. Is he right or is he just messing with me? and how can I prove this?
- (Carbon) What roles do humans play in the Carbon cycle (at least three)?
- (Nitrogen) Why is my grass always greener after a thunderstorm… even if it doesn’t rain that much?
- Cycles Review (pdf). After taking the notes, and either before or after answering the three questions, do this worksheet to check your understanding of the material. The key is posted on the Semester 2 Documents page.
On wednesday we’ll start discussion of Human impacts on the environment so you’ll need to know these cycles so we can discuss how we as humans disrupt them.
Chemistry
Please continue to work on your gas laws unit. You should be somewhere between #7-10 by the end of this week to be on a good pace. Some of you are still lagging behind…
Thank you again for your understanding and I’ll see you Wednesday. – Mr. K
Thursday 4/10
 I’ll be out today doing some professional development over at Fatima. If you need me, send a quick email. Otherwise, you are working in an open classroom at your own pace. Please be respectful of the substitute and work on something during class even if it is not something related to our class. Enjoy the great weather and have a good day!
I’ll be out today doing some professional development over at Fatima. If you need me, send a quick email. Otherwise, you are working in an open classroom at your own pace. Please be respectful of the substitute and work on something during class even if it is not something related to our class. Enjoy the great weather and have a good day!
Biology
- Biome Books – Due Monday
- Interdependence Notes – Must be completed before class tomorrow
- Interdependence Guided Practice – Can be done anytime today or tomorrow but not until notes are done
- Wolf/Moose Article – On the back of the guided practice (or the first page of the lab pdf)
- Predator/Prey Lab – Done in class on Friday. Will take approximately 30-40 minutes to complete. Lab report due Monday but can be turned in Friday
Chemistry
- Continue working on the open classroom format. As you complete activities, show the sub you have them done (check your own work online) and have him initial your work. Keep in mind it is up to you to control your learning so if you choose to cheat the system and copy work to get extra sections done it will only hurt you in the long run.
Unit 10: Gas Laws Overview

Today we’re going to begin studying Gas Laws… (and that doesn’t mean the rules on Dutch Oven or letting one go in a crowded elevator).
We’re going to try some new classroom techniques for this final unit and create an “Open” classroom. Below are the expectations of you the student and of me the teacher.
Student Values:
Unit 10: Complete the Puzzle
6th Period Chemistry – Activity Chart
Gas Laws Resource Living Document
1. Properties of Gases
- Describe and site 4 examples of how gases are different than solids and liquids at the molecular level.
- Complete each of these conversions: atm → mmHg // atm → kPa // atm → torr
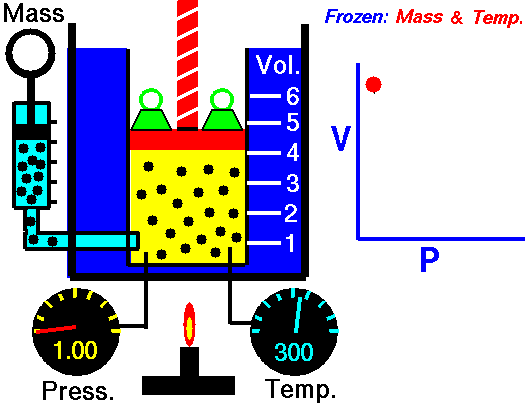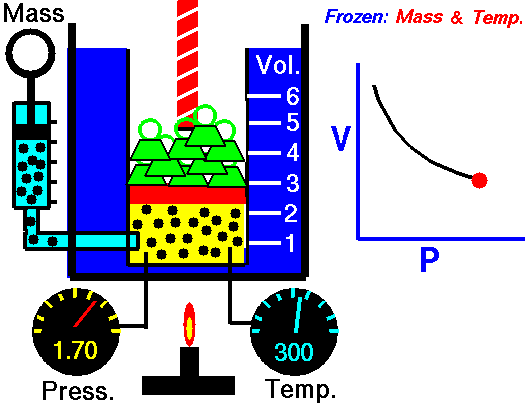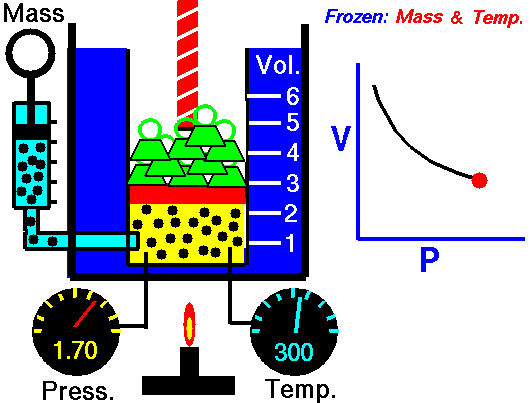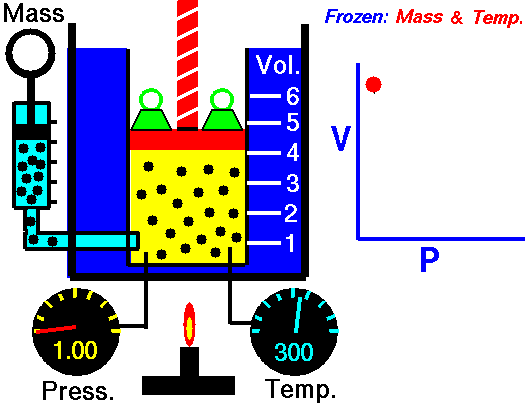
2. Boyle’s Law
- Describe Boyle’s Law
- Complete 4 problems using Boyle’s law
- Write out an explanation of how you can remember the formula for Boyle’s Law.
3. Charles Law
- Describe Charles law
- Complete 4 problems using Charles’ law
- Watch this video and write an explanation of how it proves Charles’ Law.
4. Gay-Lussac’s Law
- Describe Gay-Lussac’s law
- Before receiving completion for this section you should have: Posted at least 1 source of your information (that was not previously posted) , with your name to the Google Doc… OR… Post a question you have and/or Post a response to another students question.
5. Combined Gas Law
- Complete 4 problems using the Combined Gas Law.
- Write and submit an explanation of what the combined gas law is and why it is useful in the box below. You can see the text from other respondents by clicking this link. Combined Gas Law Responses
6. Standard Temperature & Pressure (STP)
- Complete 2 problems using the Combined Gas Law at STP.
- Write a Definition of Standard Temperature & Pressure.
7. Universal Gas Constant (R)
- Find ‘R’s value for :
- L·Torr/mol·K (L·mmHg/mol·K)
- liter·atm/mol·K
- Determine what formula is ‘R’ used in.
8. Ideal Gas Law: Volume
- Write out the formula of the Ideal Gas Law.
- Complete 4 problems finding the value of the Universal Gas constant using the Ideal Gas Law.
9. Ideal Gas Law: Moles
- Determine how many liters are equal to 1 mole of any substance at STP (#19 on the Gas Laws Calculations)
- Complete 4 problems using the Ideal Gas law.
10. Density
- Label the parts of this formula: ρ = MM·P/RT
- Complete 5 problems for density of Gases.

